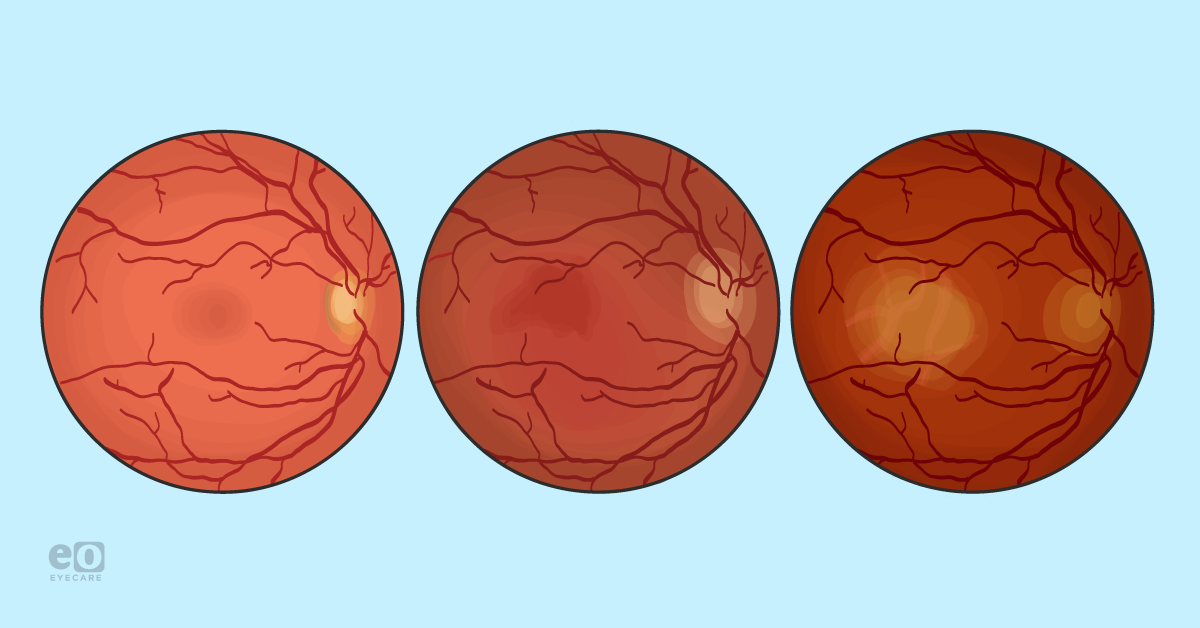
Age-related macular degeneration (AMD) is an acquired maculopathy characterized by drusen, retinal pigment epithelium (RPE) changes, retinal and choroidal atrophy, and/or macular neovascularization most often occurring in patients older than 50 years of age.
An estimated 196 million people have been diagnosed with AMD as of 2020, with that number growing to 288 million by 2040.
1 Likewise, the prevalence of advanced AMD might increase from 11.3 million in 2020 to 18.6 million in 2040, with distribution roughly split between
geographic atrophy (GA) and neovascular AMD.
2GA is considered the dry form of advanced AMD and is diagnosed when areas of outer retinal and RPE atrophy develop. Neovascular AMD, or the wet form of advanced AMD, occurs when patients develop macular neovascularization (MNV).
Since pathological neovascularization within the macula can originate from the choroidal or retinal circulation, the term choroidal neovascularization has been replaced with the less specific term, MNV.
Stages of age-related macular degeneration
For many years, the classification of AMD was based on a simplified version of the Age-Related Eye Disease Study (AREDS) severity scale, but has since been supplanted by the
Beckman Committee Criteria.
3 Understanding the high risk of vision loss associated with non-central GA, the Beckman Committee criteria labels any eye with GA as late AMD. This is in contrast to the
AREDS classification, which labeled non-central GA as intermediate AMD.
Table 1 outlines the differences between the Beckman Committee Criteria and AREDS criteria for AMD classification. Note, for the Beckman Committee Criteria, all findings must be within two disc diameters (DD) of the fovea, drusen include any drusen type and subretinal drusenoid deposits, and pigment abnormalities refer to any hyperpigmentation or hypopigmentation associated with medium or large drusen.
| AMD Stage | Beckman Committee Criteria | AREDS Criteria |
|---|
| No apparent aging changes | No drusen and pigment abnormalities | |
| Normal aging changes | Small drusen only | |
| Early AMD | | No or few small drusen |
| Intermediate AMD | Large drusen and/or pigment abnormalities | Numberous medium drusen, at least one large druse, and/or non-center involving geographic atrophy |
| Late/Advanced AMD | Neovascular AMD and/or geographic atrophy | Center involving geographic atrophy, MNV or current/past signs of MNV |
Table 1: Courtesy of the authors.
Identifying AMD and GA with OCT imaging
Table 2 features OCT findings associated with various stages of AMD and GA.
| AMD Finding | Description |
|---|
| Geographic atrophy | Clinical term used to denote areas of retinal and RPE atrophy without the presence of present/past MNV. |
| Incomplete RPE and outer retinal atrophy (iRORA) | Vertically-aligned photoreceptor/outer retinal degeneration, RPE attenuation or disruption, and increased signal transmission into the choroid. Must not qualify as cRORA. |
| Complete RPE and outer retinal atrophy (cRORA) | Vertically-aligned zone of hypertransmission of ≥250μm, zone of attenuation or disruption of RPE band of ≥250μm, and evidence of overlying photoreceptor degeneration whose features include outer nuclear layer thinning, external limiting membrane loss, and ellipsoid zone or interdigitation zone loss. Must exclude scrolled RPE or other signs of RPE tear. |
Table 2: Courtesy of authors.
Figure 1 is OCT imaging of choroidal hypertransmission (denoted by red) with vertically aligned outer retinal and RPE atrophy (denoted by yellow) in this patient with complete RPE and outer retinal atrophy (cRORA).
Figure 1: Courtesy of Daniel Epshtein, OD, FAAO.
Progression of AMD
Once clinical signs of AMD have manifested, disease progression will likely occur in essentially all AMD patients. Though it is not fully understood, it is thought that
progression to the two forms of late AMD constitutes two distinct but related paths of worsening disease. As noted in the AREDS, patients with early AMD had a 1.3% risk of developing advanced AMD within a 5-year period.
5 This number increased to up to 50% in intermediate AMD patients, depending on the severity of their clinical findings.
6GA tends to initially affect one eye before progressing to bilateral disease. Atrophy often begins extrafoveally and then expands inwards and peripherally, with a median time of 2.5 years till subfoveal involvement.
2 Patients with
multifocal lesions, large lesions at baseline, and bilateral disease are more likely to experience rapid progression and vision loss.
2While the clinical entity of combined GA and MNV is poorly studied, nevertheless, it is thought to represent an even more advanced form of AMD than either GA or MNV alone. AMD patients can progress to this combined GA and MNV stage via two clinical pathways: GA patients can develop MNV, or MNV patients can develop GA.
Prevalence of geographic atrophy progression
The rates of progression from GA to combined GA and MNV vary significantly, with studies reporting 3.4% to 49% of GA eyes developing MNV, depending on the follow-up period.
7,8 Though a rate of 49% MNV development in GA patients may seem surprising to clinicians,
histopathological studies have revealed that 33% of clinical GA patients harbor MNV.
9 This high rate of histopathological MNV in GA patients may be explained by the difficulty of detecting very mild MNV with ophthalmoscopy or the presence of nonexudative MNV. The role of GA in MNV has mostly been studied in the context of how anti-vascular endothelial growth factor (VEGF) treatment affects the rate of GA progression. In the
Comparison of Age-related Macular Degeneration Treatments Trials (CATT), 41% of MNV eyes undergoing anti-VEGF treatment demonstrated GA at the 5-year mark.
10Patients receiving monthly anti-VEGF treatment were more likely to have GA than those receiving PRN anti-VEGF treatment.10 Though still inconclusive, this data has added credence to the hypothesis that titrating anti-VEGF therapy to maintain a nonzero amount of VEGF is imperative to prevent retinal atrophy while treating exudative MNV.
Visual deficits in late AMD
The majority of visual deficits from AMD occur secondary to GA or MNV, with 90% of all AMD-related legal blindness caused by the latter.11 Due to the relatively rapid progression and foveal preference of MNV, most vision loss from MNV will be sudden, with a tendency to progress over the course of weeks to months. Vision loss associated with GA is often more insidious due to its slow progression and predominantly extrafoveal location.
Once GA has extended into the fovea, patients will experience near-absolute central scotomas. The median time from extrafoveal to foveal GA is approximately 2.5 years, and during this time, patients may complain of visual function deficits even though their visual acuity is preserved. Tasks such as reading or those that require visual scanning are particularly affected, usually to a greater extent than expected from visual acuity measurements.
Even in patients with small GA lesions, visual function in reduced illumination can be affected, leading to vague, subjective complaints of poor night vision. Whereas vision loss from MNV may be reversed with
anti-VEGF therapy, all visual deficits caused by GA are irreversible.
Geographic atrophy imaging biomarkers
Here are our quick takeaways and assessments for each of these imaging studies:
- Color fundus photography (CFP): An imaging modality for documenting large drusen burden and drusen regression, which have both been linked to a higher risk of GA development.
- OCT: Signs such as subretinal drusenoid deposits and hyper-reflective foci can also incur a higher risk of conversion from intermediate AMD to GA. The presence of iRORA is considered an indication that the atrophic process towards GA has already begun and should be screened for in every AMD patient.
- Fundus autofluorescence (FAF): Can be used for both detection and prognostication purposes in GA. Atrophic areas will appear as hypoautofluorescent or dark. Often, these areas will be surrounded by hyperautofluorescence, which often indicates the direction of GA expansion. Certain hyperautofluorescent phenotypes such as banded pattern, diffuse pattern, and diffuse-trickling pattern have been associated with a faster rate of GA progression.
Current studies on treatments for GA
Phase 3 studies from Apellis Pharmaceuticals
The
OAKS and DERBY trials enrolled 637 and 621 patients, respectively; 419 patients were randomized to pegcetacoplan (APL-2) monthly, 420 to pegcetacoplan every other month, and 417 were randomized to either sham monthly or sham every other month.
12The primary endpoint was at 12 months, evaluating the change in total GA lesion area on FAF. The 24-month secondary endpoints evaluated best-corrected visual acuity (BCVA), reading speed, microperimetry, as well as lesion growth. Patients had to be at least 60 years old, BCVA 20/320 or better, and GA lesions with or without subfoveal involvement were allowed. The presence of MNV in the study eye was exclusionary, while the presence of MNV in the fellow eye was not.13
There was a 21% reduction in GA lesion growth at 2 years for the pegcetacoplan monthly group compared to sham. Further, there was a 17% reduction in GA lesion growth at 2 years for the Pegcetacoplan every other month group compared to sham. These percentages were statistically significant.13
When comparing the study eye versus the untreated fellow eye, monthly treatment showed a 19% difference, whereas every other month treatment showed a 16% difference. These percentages were statistically significant.13
In the non-subfoveal subgroup analysis, monthly treatment with pegcetacoplan (158 patients) resulted in a 26% reduction in GA lesion growth, whereas every other month treatment (155 patients) resulted in a 22% reduction in GA lesion growth. These percentages were statistically significant.13
Note: Apellis Pharmaceuticals received FDA approval for Syfovre (pegcetacoplan injection) 15mg/0.1 mL in February 2023.
Phase 3 studies from Iveric Bio
In the
GATHER1 trial, in part one, 77 patients were randomized to avacincaptad pegol (ACP) 1mg, ACP 2mg, or sham, given monthly. In part two, 209 patients were randomized to ACP 2mg, ACP 4mg, or Sham, given monthly. The primary analysis was at month 12, and descriptive analysis was at month 18.
14GATHER1 illustrated a 27.4% reduction in mean change in GA area from baseline compared to sham. Additionally, the trial showed a 35.4% reduction compared to sham in the mean rate of GA growth from baseline. In totality, these percentages were statistically significant.14
Switching to the
GATHER2 trial, 448 patients were randomized to ACP 2mg vs. sham, given monthly. After 1 year, half of the ACP 2mg group was randomized to continue with monthly injections and the other half received injections every other month. The sham group continued with monthly sham injections.
14GATHER2 showed a 14.3% reduction in mean change in GA area from baseline compared to sham. Further, this trial demonstrated a 17.7% reduction compared to sham in the mean rate of GA growth from baseline. Similar to GATHER1 results, these percentages were statistically significant.14
Of note, both studies involved patients above the age of 50, with BCVA between 20/25 and 20/320, with GA within 1500 microns from the foveal center point, but not involving the foveal center.14
Note: Iveric Bio, An Astellas Company, received FDA approval for IZERVAY (avacincaptad pegol intravitreal solution) 2mg (0.1mL of 20mg/mL solution) in August 2023.15
Rates of ocular inflammation
While there were no cases of occlusive vasculitis or retinitis reported in the DERBY and OAKS study data, there were 28 cases of intraocular inflammation out of 11,736 pegcetacoplan injections.
All of these cases were evaluated by neuro-ophthalmologists, with three patients found to have
ischemic optic neuropathy with discs at risk and multiple systemic risk factors. Four cases of inflammation reported in 2018 were due to drug impurity, and infectious endophthalmitis rates were 1 in 3,000 injections.
16,17There was one case of intraocular inflammation in the GATHER1 trial. In GATHER2, there were no cases of intraocular inflammation,
endophthalmitis, or ischemic optic neuropathy in study eyes treated with ACP 2mg at 1 year.
18Rates of macular neovascularization
Both treatments for geographic atrophy APL-2 and ACP showed an increased incidence of conversion to wet macular degeneration, characterized by the
formation of MNV.
13,14The 2-year data from DERBY and OAKS found rates of MNV of 11.9% in the group treated monthly with APL-2, 6.7% in the group treated every other month with APL-2, and 3.1% in the Sham group.13,14
The 1-year data from GATHER1 illustrated rates of MNV of 9.6% in the ACP 4mg group, 9.0% in the ACP 2mg group, and 2.7% in the sham group. Subsequently, the 1-year data from GATHER2 showed rates of MNV of 6.7% in the ACP 2mg group and 4.1% in the sham group.13,14
Patient selection for GA treatments
Most of my (Deep Parikh, MD) initially treated patients tend to be monocular due to subfoveal geographic atrophy in one eye and have extrafoveal geographic atrophy in the other eye. These patients tend to be the most motivated to prevent further vision loss in the eye with extrafoveal GA and understand how quickly GA can progress.
As a reminder, it can take, on average, up to 2.5 years for
GA lesions to start impacting central vision and, thus, our patients' ability to read, drive, and see their loved ones' faces.
19 So, as doctors and patients have gotten more comfortable with this novel medication (Syfovre), its use is becoming more commonplace. More and more patients are starting to request treatment for GA, including those with extrafoveal GA in both eyes.
When to refer GA patients to retina specialists
I think the ideal patients for
optometrists to consider referring to the retina specialist include patients with intermediate dry macular degeneration with the early start of geographic atrophy, caught on OCT. Additionally, primary eyecare providers may want to consider early referral to a retina specialist for patients who have factors that suggest their condition might progress rapidly.
This includes patients with multifocal GA lesions, extrafoveal lesions, subretinal drusenoid deposits, increased autofluorescence patterns at the junctional border of GA, including diffuse or banded patterns, OCT demonstrating findings of nascent GA, and smokers.20
OCT and near-infrared reflectance (NIR) imaging on the OCT can be the most useful methods to detect the
development of GA. Signs include loss of photoreceptor and RPE layers suggestive of outer retinal atrophy as well as light hyper-transmission and increased choroidal reflectivity below Bruch’s membrane.
20 FAF is an invaluable tool but is not as readily available in the ophthalmic community outside of retina practices.
Of note, both Apellis and Iveric Bio’s studies relied on FAF to track GA growth.13,14
GA treatment case study
A 79-year-old man reported rapid deterioration in the vision of his right eye over the past 2 years due to advanced
dry macular degeneration. Visual acuity was 20/60 eccentrically in the right eye and 20/25 in the left eye. After discussing the literature findings and reviewing risks, benefits, alternatives, and contraindications, he elected to undergo treatment with intravitreal complement inhibition monthly to help preserve his vision in the left eye.
Figure 2, top row from left to right, NIR and OCT show foveal involvement, and NIR and OCT OS show slight sparing of the fovea. Bottom row, microperimetry demonstrating central scotoma greater in the right eye than the left eye.
Figure 2: Courtesy of Deep U Parikh, MD.
Treating comorbid GA and MNV
While GA and MNV are generally considered to be two distinct entities, they can happen simultaneously in the same eye. It may be that they are actually two different diseases, as was previously thought, or that both of these coexist within the spectrum of a single disease.21
Interestingly, GA and MNV are both associated with the same common genetic variants: CFH and ARMS2 genes. They both also present with drusen and subretinal drusenoid deposits, and can each occur simultaneously with a frequency that is likely underestimated. Consequently, there is relatively strong evidence suggesting these diseases might overlap in their pathophysiology.22
Older histopathological studies reported on rates of combined GA and MNV.
Sarks et al. found that 15 of 26 eyes with a clinical diagnosis of GA had subclinical MNV on histology, while conversely,
Green et al. found that 22 eyes of 63 patients with MNV had areas of GA on histology. In
another study, 86 of 760 eyes with a diagnosis of AMD were found to have both MNV and GA on post-mortem analysis. It is likely that rates of patients with combined GA and MNV are underestimated.
21Conversion from MNV to GA
There is some evidence that treatment with anti-VEGF agents may increase the rate of GA formation.
Grunwald et al. reported that of 1,185 patients receiving anti-VEGF treatment, 10.1% (120 patients) developed GA within the first year, and an additional 3% (36 patients) developed GA within the second year. Fortunately, they found that the GA developed in areas of MNV involvement, and this GA was clinically similar to de novo GA and grew at similar rates.
21,22The CATT Study illustrated that 20% of patients with
wet AMD undergoing anti-VEGF treatment go on to develop GA after 2 years, which doubled to 41% in 5 years. This was found to result in severe vision loss. Authors concluded that where previously pre-anti-VEGF era patients had fibrotic scars, now they may be evolving into atrophic scars with even worse vision with anti-VEGF therapy.
23,24Conversion from GA to MNV
Rates of development of MNV in GA patients vary in the literature.
Sunness et al., in the pre-anti-VEGF era, reported that of 152 patients diagnosed with GA in at least one eye, at 2 years, 6% developed MNV, and at 4 years, 17% developed MNV in the study eye. The eyes that developed MNV had more vision loss than those with only GA. The eyes that had MNV sometimes had an evanescent appearance that left a final appearance of an enlarged area of GA.
25The
Beaver Dam Study found that 10.9% (6 of 55) of patients with GA progressed to MNV after 5 years. They found that rates of progression were higher if MNV was present in the fellow eye.
26Even more recent evidence from the phase 3 studies from Iveric Bio and Apellis Pharmaceuticals suggests that treatment with anti-complement agents further increases rates of MNV formation, supporting the notion that both conditions may represent different components of the same disease spectrum.13,14
Secondary MNV in the setting of treatment for geographic atrophy
But the real question remains—how bad is choroidal neovascularization in the setting of treatment for geographic atrophy? Well, recent case reports and studies show that the MNV formed in the setting of geographic atrophy responds well overall to anti-VEGF agents. Researchers have coined the term secondary MNV, referring to this type of MNV, versus de novo or primary MNV in
wet AMD.
Siedlecki et al. looked at rates of GA growth before and after diagnosis of a secondary MNV treated with anti-VEGF therapy. Fortunately, this study showed that GA enlargement was not significantly affected by a secondary MNV in the anti-VEGF era. They also showed that there was no difference in the rate of GA enlargement between eyes that developed MNV and the fellow eye that did not develop MNV.
23 Similarly, other studies from
Amaro et al. and
Casalino et al. show an overall favorable response to anti-VEGF for secondary MNV in GA patients with stable resulting VA. In the study from Casalino et al., it was demonstrated that the same rate of progression in patients who had been treated for MNV was similar to those who did not have MNV.
27,28However, in one contrasting study,
Querques et al. showed that although the MNV responded very well to the anti-VEGF treatment, the area of GA actually enlarged in size. Although even in their study, the overall resulting VA remained stable, which was limited at baseline in these patients due to the pre-existing GA lesion.
29Final thoughts
We know from the wet AMD literature and our experiences since 2005 that anti-VEGF agents are effective, and we know that the earlier we catch and treat MNV, the better the patient does. Fortunately, our technology and understanding today have improved even further, and we can monitor patients with multiple imaging modalities, including OCT,
fluorescein angiography (FA), FAF, and even
OCT angiography (OCTA), allowing us to identify earlier these different types of MNV and at various stages.
Further, the monthly follow-ups and treatments required for GA patients also give us the ability to monitor patients more closely, enhancing our ability to detect MNV very early in the process and thus initiate treatment early.
As our understanding expands and as we get more information on treating and managing patients that develop secondary MNV in the setting of undergoing treatment for GA, we will further be able to characterize these as type 1 (occult, below the RPE) versus type 2 (classic, above the RPE) and exudative versus nonexudative or quiescent MNV.
Some new data suggests that not all MNV is bad, and there may be a beneficial role for some type 1 MNV that exist in a trophic state, are nonexudative, and may actually provide metabolic support for the overlying RPE and retina.22 Of course, more data will be needed, and further analysis on patients treated with complement inhibitors for GA who go on to develop MNV.
Early reports seem favorable. It’s possible that combined complement inhibitor and anti-VEGF treatments may exist in the future, as well as different treatment algorithms such as treating GA and additionally giving anti-VEGF prophylactically q6 months, but studies will need to look into these different strategies.
The future is very bright for treating retinal diseases, giving hope for our patients suffering from AMD, especially those with GA.