However, the vast choices available can sometimes be overwhelming, and it is important to provide pre-surgical patients with a clear understanding of what cataract surgery is, and how we can improve and enhance their vision with new and advanced technology—
intraocular lens implants.
This article outlines some of the more common IOL technologies available and their specifics.
Why is intraocular lens implantation necessary?
An intraocular lens, with respect to cataract surgery, is an artificial lens that is placed into the eye at the time of cataract extraction.1-3 The IOL replaces the eye’s natural lens that is removed during the surgery. The natural crystalline lens inside the eye bends (refracts) light to allow for clear images.1-3 However, as a cataract forms (i.e. the lens becomes cloudier), images seen become gradually hazier and dimmer in nature.
Therefore, cataract surgery removes this natural cloudy lens and replaces it with a clear IOL to help enhance and improve vision.
1-3 Cataract extraction without implantation of an IOL (i.e.,
aphakia) can lead to very blurry vision, and the need for incredibly high prescription (“coke-bottle”) eyeglasses or contact lenses, which can be very difficult to tolerate.
Therefore, standard-of-care cataract surgery always includes intraocular lens implantation during the procedure. In general,
routine cataract surgery involves IOL implantation within the capsular bag complex; less commonly the IOL may be implanted in the ciliary sulcus or anterior chamber.
1-3IOLs come in different focusing powers—similar to prescription eyeglasses or contact lenses. The correct power IOL for the eye is determined by extensive measurements and calculations performed at the
time of cataract evaluation.
1-3 Most IOLs are composed of acrylic or silicone materials—these are biocompatible and are not rejected by the body once implanted. Some IOLs are also coated with a special UV-blocking material to help protect from harmful UV rays.
1-3 Below we review the different types of IOLs available.
IOL Categories
Monofocal
The most common type of lens used with cataract surgery is a monofocal IOL. Monofocal means “one focal point” or “one focus”. It is therefore set to focus for distance, intermediate, or near only (but not all three).1-4 Most patients who choose monofocal IOL’s choose distance correction, and therefore use glasses for reading, working on a computer, and other near activities postoperatively.
Alternatively, patients may also choose to have monovision, where one eye (typically the dominant eye) is corrected for distance, and the other eye is corrected for near, providing a range of vision from near to distance.1-5 It is recommended that patients trial monovision with contact lenses prior to making the decision to have cataract surgery with monovision to ensure they can tolerate it. Not all patients like monovision and they may have altered depth perception that could affect their ability to safely navigate walking up and down stairs.1-5
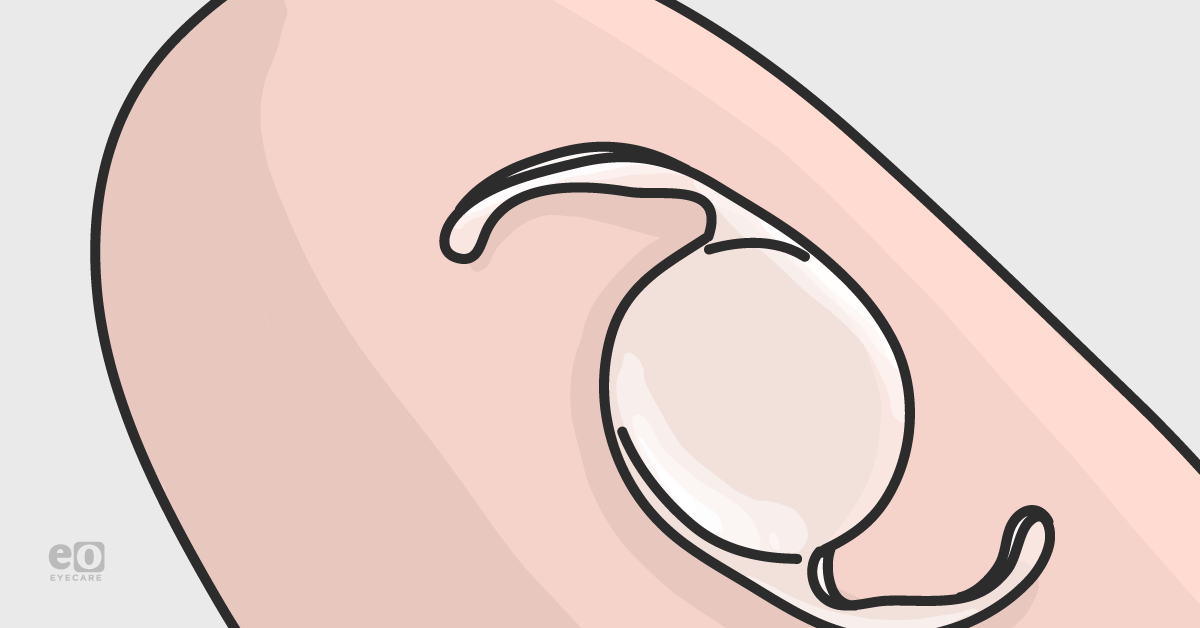
An example of what monofocal IOLs look like is seen in the figure below. Figure 1a (far left) specifically shows a 1-piece monofocal IOL (this is implanted in the capsular bag), Figure 1b (center) shows a 3-piece monofocal IOL (may be implanted in capsular bag or ciliary sulcus), and Figure 1c (far right) shows an example of an anterior chamber (ACIOL) IOL.
Figure 1a. Single-piece IOL6 - Figure 1b. 3-piece IOL7 - Figure 1c. ACIOL8
- Varieties (There are several brands available in the USA: Alcon (Fort Worth TX), Johnson & Johnson Vision(Jacksonville FL), Bausch + Lomb (Bridgewater NJ), BVI (Waltham, MA))
- Single-piece IOLs: this is implanted in the capsular bag only. 1-3 An illustration of a single-piece IOL is seen in Figure 1a.
- 3-piece IOLs: typically has an acrylic or silicone optic with thin PMMA, polypropylene, or polyvinylidene fluoride (PVDF) haptics that may be implanted in the capsular bag, ciliary sulcus, or scleral fixated. 1-3, 6 An illustration of a 3-piece IOL is seen in Figure 1b.
- Anterior chamber IOLs (ACIOL): these are 1-piece IOLs meant to be implanted in the anterior chamber and rest on the iris. It is essential to create a peripheral iridotomy/iridectomy at time of insertion to prevent pupillary block. These IOLs cannot be folded at time of insertion and typically require a 6mm incision size that is sutured closed.1-3
- Expected Visual Acuity: Most patients who choose monofocal IOLs choose distance correction, and therefore use glasses for reading, working on a computer and other near activities postoperatively. Alternatively, patients may also choose to have monovision, where one eye (typically the dominant eye) is corrected for distance, and the other eye is corrected for near, providing a range of vision.1-3
- Mechanism of Action: Provides “one focal point” or “one focus”. It is therefore set to focus for distance, intermediate or near only (but not all three). The focal point, and therefore IOL power, is calculated and chosen by the surgeon and patient prior to surgery.1-3
- Allows maximum amount of light through to the retina with minimal light energy lost in comparison to multifocal IOLs1-3
- Ideal Patient Candidates: All patients are considered good candidates for monofocal IOLs; these lenses have minimal side effects and generally provide the sharpest vision and contrast sensitivity.1-3
- Side effect profile: Minimal risk of glare/haloes
- Standard risk of visual aberrations with IOL decentration or other surgical complications
- Relevant FDA data: IOLs should be used as specified (e.g., no single-piece IOL’s in the sulcus!)
Multifocal
Multifocal (MFIOL) IOLs provide focusing for at least two focal points (e.g. distance and near, or distance and intermediate).1-4,10 These IOLs decrease the need for glasses postoperatively by providing good vision across a range of distances. The lens has special zones set at different powers to allow for an enhanced range of vision.10,11 As many MFIOLs are manufactured with a diffractive ring design12 (Figure 2), it is possible that some patients may experience side effects, such as glare, haloes, or starbursts around bright lights—most commonly while driving at night.13
In addition, in an attempt to provide a full range of vision, the diffractive ring design can lead to a compromise in contrast sensitivity—or how sharply images are seen.
10-15 Therefore, it is usually not recommended to implant MFIOLs in patients with advanced macular degeneration,
glaucoma, and other retinal diseases that already cause compromised visual acuity.
10-15Technology surrounding multifocal IOLs has advanced significantly, and it is much less common for patients to experience severe visual adverse effects from newer lenses, however, it is certainly something to be aware of and imperative to include in the consent process.
Figure 2. Diffractive IOL Design (12)
Traditionally, MFIOLs in the USA were bifocal lenses—permitting vision at distance and near, or distance and intermediate but not all three focal points (distance-intermediate-near).10,11,14,15 Though these IOLs function well, some patients realized that they were “missing part of their vision” (i.e. in bilateral distance-near or bilateral distance-intermediate IOL implantation), and this lead to surgeons using a mix-and-match technique to help provide patients with a full range of vision from near to intermediate to distance.10,11,14,15
This would be done by placing a “distance-near” MFIOL in one eye and a “distance-intermediate” IOL in the other. Though this worked well in some patients, a true “full range of vision” from an IOL did not come about until a trifocal lens was introduced in the US in 2019 (PanOptix, Alcon, Fort Worth TX).10,11,14-16
Trifocal IOLs provide patients with vision at near, intermediate and distance.15-17 There is one trifocal IOL FDA approved in the US at this time (PanOptix) and many patients have been incredibly happy with its visual results.15-17 However, it is important to note that it is still possible to have glare, halos, and starbursts with these IOLs, although the PanOptix lens has reportedly less incidence of this compared to the early iterations of bifocal IOLs.15-17
- Varieties
- Bifocal: Traditionally, MFIOLs in the USA were bifocal lenses—permitting vision at distance and near, or distance and intermediate but not all three focal points (i.e., distance-intermediate-near)1-5,14, 16-17
- Available in Toric and Spherical versions
- Trifocal: These IOLs provide patients with vision at near, intermediate and distance. There are two trifocal IOLs FDA approved in the US at this time (PanOptix, Alcon, Fort Worth TX and TECNIS Synergy, Johnson & Johnson Vision, Jacksonville FL)1-5, 14, 16-17
- Note: The Synergy IOL is a combination of a multifocal IOL and extended depth of focus technology, thereby combining two mechanisms to provide an extended range of vision.
- Available in toric and spherical versions
- Expected Visual Acuity: These IOLs decrease the need for glasses postoperatively by providing good vision across a range of distances. However, patients should be advised that very fine work, or near work done at a short distance from their face may still require reading glasses (generally at a distance closer than 40cm).1-3,4-5,10-16
- Mechanism of Action: Multifocal (MFIOL) IOLs provide focusing for at least 2 focal points (e.g. distance and near, or distance and intermediate). The lens has special zones set at different powers to allow for the enhanced range of vision.1-3, 4-5,10-17
- Diffractive optics (in brief): Bifocal and Trifocal IOLs generally work via diffractive optics which take advantage of the wave-nature of light and constructive as well as destructive interference. Waves of light also undergo diffraction when they interact with boundaries and sharp edges (i.e., rings on an IOL optic)—this causes the light to bend around the edges and travel in a different direction. In diffractive MFIOLs, a specifically shaped structure is usually placed on the lens surface that intentionally induces diffraction so that waves exiting the lens will have constructive interference at 2 (or more) distinct foci.18 An illustration of diffractive IOL technology is seen in Figure 3.
- Ideal Patient Candidates: Due to potential side effect profile (i.e., glare, haloes, decreased contrast sensitivity), MFIOLs are usually not recommended in patients with advanced macular degeneration, glaucoma, corneal dystrophies and other ocular diseases that already cause compromised visual acuity. 1-3, 4-5,10-17
- Side Effect Profile: As many MFIOLs are manufactured with a diffractive ring design, it is possible that some patients may experience side effects, such as glare, haloes, or starbursts around bright lights—most commonly while driving at night. In addition, in an attempt to provide a full range of vision, the diffractive ring design can lead to a compromise in contrast sensitivity—or how sharply images are seen. Technology surrounding multifocal IOLs has advanced significantly and it is much less common for patients to experience severe visual adverse effects from newer lenses, however, it is certainly something to be aware of and imperative to include in the consent process.1-3, 4-5,10-17
- Relevant FDA Data: Multifocal IOLs (bifocal and trifocal varieties) have been FDA approved for surgically naïve (e.g. no prior LASIK, PRK, RK, other corneal surgeries, etc) eyes. Certain IOLs have been studied in post-refractive surgery patients (phase IV, post-market studies), however, caution is advised in any patients with significant corneal pathology, or disease states that can predispose to poor visual outcomes (e.g., severe pseudoexfoliation, glaucoma, macular degeneration, etc).1-3, 4-5,10-17
Figure 3: Diffractive IOL technology
Extended depth of focus:
Extended depth of focus (EDOF) IOLs aim to provide patients with a functional range of vision (usually excellent intermediate to distance with reasonably good near vision), with a similar visual disturbance profile to a monofocal lens.1,19,20 This is in comparison to MFIOLs, which have specific focal points for near, intermediate, and distance built into their design.1,10,11,19,20 In this way, this subset of IOL’s can offer patients some degree of spectacle independence, with a significantly lower incidence of visual disturbances.1,10,11,19,20
Two different EDOF IOLs are available on the US market at this time: the
TECNIS Symfony IOL (Johnson-Johnson Vision, Santa Ana CA) as well as the newer
Vivity IOL(Alcon, Fort Worth TX). Both IOLs are manufactured using different optical principles and therefore may offer some advantages in certain patient populations. For example, the Vivity IOL does not utilize diffractive optics in its design, so theoretically it has a lessened
incidence of glare and haloes postoperatively—side effects that may still be possible, even with EDOF lenses.
20An example of the Vivity IOL is seen in Figure 4.20
Figure 4: Vivity Extended Vision IOL (20)
- Varieties
- TECNIS Symfony IOL (Johnson & Johnson Vision, Jacksonville, FL)
- Vivity Extended Range of Vision IOL (Alcon, Fort Worth TX)
- Expected Visual Acuity: Extended depth of focus (EDOF) IOLs aim to provide patients with a functional range of vision (usually excellent intermediate to distance with reasonably good near vision), with a similar visual disturbance profile to a monofocal lens.1,2,20 Expected acuity for distance and intermediate activities (intermediate meaning working on a laptop, seeing the car dashboard) is excellent; near work such as reading articles/text on a cell phone may still require a light pair of reading glasses.1,2,20
- Mechanism of Action:
- TECNIS Symfony IOL: extends depth of focus through a combination of effects from its echelette design, reduced chromatic aberration and negative spherical aberration.1,2,5,11,20
- Available in toric and spherical versions
- Vivity IOL: Does not utilize diffractive optics in its design, so theoretically it has a lessened incidence of glare and haloes postoperatively.1,2,5,11,20 It has patented X-Wave™ technology that simultaneously stretches and shifts the image/light coming into the eye (i.e., it never splits light) to provide a continuous extended focal range.1,2,20
- Available in toric and spherical versions
- Ideal Patient Candidates: Due to a side effect profile similar to a monofocal IOL, it may be possible to implant these IOLs in post-refractive surgery patients, as well as other eyes that may be at risk for increased higher order aberrations. 1,2,6,8,13 However, patients should still be carefully counseled regarding side effects; ideal patients still remain those with excellent visual potential, minimal-to-no additional ocular pathology, and those willing to use reading glasses for some near activities.
- Side Effect Profile: It is still possible to experience starbursts (more notable with the Symfony IOL) as well as glare and haloes with the EDOF IOLs (although to a lesser degree than with traditional bifocal IOLs).1,2,5,11,20 Patients need to be counseled that near vision with EDOF IOLs is typically not as sharp in comparison to trifocal IOLs.1,2,5,11,20
- Relevant FDA Data: Like MFIOLs, EDOF lenses were also approved to treat presbyopia in cataract surgery patients. FDA trials did not focus on post-refractive surgery patients and those with underlying/additional ocular pathologies. Careful consent and discussion should be had with any patient considering an EDOF lens for cataract surgery.
Toric
Toric IOLs correct astigmatism, which is part of the eyeglass prescription.1-3,20,21 If astigmatism is not accounted for/corrected at the time of cataract surgery there is a high chance an eyeglass (or contact lens) prescription for both distance and near will be required postoperatively.21 Toric IOLs are available in monofocal, multifocal and EDOF varieties. Toric lenses typically have markings that are aligned for the patient’s axis of corneal astigmatism.
An example of this is seen in Figure 5.22
Figure 5. Toric IOL with astigmatic markings (22)
- Varieties: Toric IOLs are available in monofocal, multifocal and EDOF varieties across several different brands 1,2, 22-25
- Alcon: can correct up to 4.00D of astigmatism (may vary with certain MFIOL/EDOF versions) 1,2,17,19-21
- Tecnis (Johnson & Johnson Vision): can correct up to 3.60D of astigmatism (may vary with certain MFIOL/EDOF versions) 1,2,22-25
- Bausch + Lomb: offers a slightly wider range of astigmatism correction (up to 4.50D of astigmatism) 1,2,22-25
- Expected Visual Acuity: As long as the IOL is positioned appropriately, vision is expected to be excellent. However, it is important to note that toric IOL rotation out of planned position can lead to suboptimal visual results.1,2,22-25 In fact, for every one degree of error in toric IOL alignment, there is a 3.3% decrease in correction of astigmatism (i.e., if a toric IOL Is misaligned by 10 degrees, this equates to a 33% loss of astigmatic correction).1,2,22-25
- Mechanism of Action: Toric IOLs correct astigmatism, which is part of the eyeglass prescription. If astigmatism is not accounted for at the time of cataract surgery there is a high chance an eyeglass (or contact lens) prescription for both distance and near will be required postoperatively. Toric lenses typically have markings that are aligned for the axis of corneal astigmatism.1,2,22-25
- Ideal Patient Candidates: Any patient with significant astigmatism can have a toric IOL implanted at the time of cataract surgery; it is important to consent patients about the (albeit low) risk of IOL rotation. Patients with too little or too much astigmatism that cannot be corrected by a toric IOL should be informed as such.1,2,22-25
- Side Effect Profile: Side effects related to toric IOL malrotation (or incorrect power) are usually blurry and/or distorted vision.
- Relevant FDA Data: Toric IOLs were approved to treat specific ranges of corneal astigmatism. Patients with astigmatic measurements outside of these ranges should be educated regarding their candidacy and consider other options for astigmatism correction.1,2,22-25
Accommodative
Accommodation is the process by which the eye changes optical power/focus to maintain a clear image at varying distances.26 Over time, the ability to accommodate decreases (presbyopia), and it is more difficult to focus at near.26 Accommodative IOLs move or change shape within the eye, allowing for focus at different distances. These IOLs are designed to function much in the same way as the natural lens.26-28 Although there have been several accommodative IOLs in the US market, they are not widely used due to limited accommodative response and unpredictable refractive outcomes.
- Varieties:
- Expected Visual Acuity: These IOLs are designed to provide good distance vision as well as some near, but near acuity may vary from patient to patient and eye to eye depending on accommodative response.1-3,5,29
- Mechanism of Action: Accommodative IOLs move or change shape within the eye, allowing for focus at different distances. These IOLs are designed to function much in the same way as the natural crystalline lens; they change power in response to ciliary muscle contraction.1-3,5,29
- Ideal Patient Candidates: As this is a monofocal IOL by nature, there are few limitations in its use—patients should understand that range of vision provided may be limited (i.e., not the same range or depth of focus as provided by trifocal IOLs).1-3,5,26,29 In addition, as the Crystalens optic is made of silicone (as opposed to most others with acrylic optics), it should be avoided in patients with asteroid hyalosis, and those who may require retinal surgery with silicone oil injection.1-3,5,26,29
- Side Effect Profile: These IOLs are not as widely used due to limited accommodative response and unpredictable refractive outcomes in comparison with other presbyopia-correcting lenses.
- Relevant FDA Data: The only accommodative IOL that has been FDA approved in the US is the Crystalens IOL.1-3,5,26,29
There are some new accommodating IOLs that may appear on the market soon:
The Atia Vision Lens (Atia Vision, Campbell CA): This IOL features a modular, 2-part design, where the back part of the device is a shape-changing implant that is intended to mimic the mechanism used in natural lens accommodation.
27,28 This part maintains direct contact with the capsular bag.
27 The front (second) section of the device is an exchangeable optic (lens) that is used to address the specific refractive needs of the patient.
27 Early preliminary studies have demonstrated this device to show accommodation within the eye. If successful, this IOL has the potential to restore a full range of functional vision to patients.27
The prototype for the Atia device is shown in Figure 6.27
Figure 6: Atia accommodative lens device (27)
The Opira Lens (ForSight Vision6, Menlo Park, CA): This IOL is a dynamic, shape-changing lens designed for placement in the sulcus with haptic fixation within the capsulorhexis.
27 This is in contrast with most other types of IOLs which are usually placed entirely behind the anterior capsulorhexis and in the capsular bag.
27 An image of the Opira lens prototype is seen in Figure 7.27
Figure 7: Opira accommodative IOL (27)
Light adjustable lens:
The first of its kind, the
Light Adjustable Lens (LAL, RxSight Aliso Viejo CA) is FDA approved and designed for the treatment of residual refractive error (i.e. eyeglass prescription) that may be present following cataract surgery.
30,31 The LAL and corresponding Light Delivery Device are indicated to allow for small adjustments to be made to the IOL power following cataract surgery so that the patient will have better vision when not using glasses.
30,31Until the development of the LAL technology, refractive errors that are common following cataract surgery could only be corrected with glasses, contact lenses, or refractive surgery (e.g. LASIK, PRK). This system provides a new option for certain patients that allows the physician to make small adjustments to the implanted IOL during several in-office procedures after the initial surgery to improve visual acuity without glasses.30,31 The RxSight IOL is made of a unique material that reacts to UV light, delivered by the corresponding light delivery device, 17-21 days post-surgery.30,31
Patients receive 3-4 light treatments over a period of 1-2 weeks, each lasting 40-150 seconds, depending on the amount of adjustment needed.30,31 The patient must also wear special UV-blocking eyeglasses from the time of cataract surgery to the end of light treatments to protect the new lens from UV light in the environment.
Figure 8 demonstrates the LAL as well as its theorized mechanism.31
Figure 8: Light Adjustable Lens before and after UV treatment (31)
- Varieties: The first of its kind, the Light Adjustable Lens (LAL, RxSight Aliso Viejo CA) is FDA approved and designed for treatment of residual refractive error that may be present following cataract surgery.30-32
- Expected Visual Acuity: Until the development of the LAL technology, refractive errors that followed cataract surgery could only be corrected with glasses, contact lenses, IOL exchange or refractive surgery.30-32 This system provides a new option for certain patients that allows the physician to make small adjustments to the implanted IOL during several in-office procedures after the initial surgery to improve visual acuity.30-32
- Mechanism of Action: The RxSight IOL is made of a unique material that reacts to UV light, which is delivered by the corresponding light delivery device, 17-21 days post-surgery.30-32 Patients receive 3-4 light treatments over a period of 1-2 weeks, each lasting 40-150 seconds, depending on the amount of adjustment needed.30-32 The patient must also wear special UV-blocking eyeglasses from the time of cataract surgery through the end of light treatments to protect the new lens from external UV light sources.30-32
- Ideal Patient Candidates: As this IOL is silicone, it is usually not recommended in patients with potential for retinal surgery with silicone oil.30-32 Patients with an adequate understanding of the IOL mechanism of action, as well as UV treatments and need for avoidance of UV exposure postoperatively are good candidates.30-32 Certain patients who may have issues with photosensitivity should not be considered for the LAL.30-32
- Side Effect Profile: It is important to discuss potential side effects of the LAL with patients. These include:
- Unpredicted change in vision due to ocular exposure to sunlight before the LAL power is “locked in” – this may necessitate IOL explantation30-32
- Transient or permanent erythropsia/color vision anomaly due to UV treatment30-32
- UV induced retinal changes30-32
- Relevant FDA Data: The LAL and corresponding Light Delivery Device are indicated to allow for small adjustments to be made to the IOL power following cataract surgery to improve vision and avoid the need for spectacle correction.30-32
New technologies on the horizon
Pin-hole IOL: A novel type of IOL, the pinhole IOL Is based on the stenopeic principle, which allows small central rays to enter the eye and eliminates diverging rays, thus reducing the circle of blur on the retina.
33,34 As a result of this, depth of focus is increased, providing patients with good distance and near vision.
33,34 Currently, two types of pinhole-based IOLs exist, though neither is yet approved for use in the USA:
IC-8 IOL (Acufocus, Irvina CA), and
Xtrafocus IOL (Morcher, Germany: Figure 9).
35-40 Though both of these IOLs have shown to increase depth of focus, and can provide this in patients who may not be good candidates for MFIOLs, these IOL’s can have a disadvantage, such as decreased brightness, decreased visual field and decreased optimal visual acuity.
35-40 In addition, patients with certain eye conditions, such as central corneal scarring, large pupils (>6mm),
severe glaucoma, severe macular pathology are not good candidates for pinhole IOLs.
35-40Figure 9: Left: IC-8 Pinhole IOL (32) Right: Xtrafocus Pinhole IOL (33)
We are very fortunate to be part of these ongoing innovations in the field of cataract surgery. With all of the options available, it is possible to significantly improve our patients’ vision. A thorough understanding of the technologies available as well as the ability to explain these options to patients will enhance your practice and allow for continuous growth as new technologies become available.
References:
- Werner L. Intraocular Lenses: Overview of Designs, Materials, and Pathophysiologic Features. Ophthalmology. 2020 Jun 30:S0161-6420(20)30626-6. doi: 10.1016/j.ophtha.2020.06.055.
- Boyd K. IOL Implants: Lens Replacement After Cataracts. American Academy of Ophthalmology. 2019. https://www.aao.org/eye-health/diseases/cataracts-iol-implants
- Lundstrom M, Barry P, Henry Y, Rosen P, Stenevi U. Evidence-based guidelines for cataract surgery: guidelines based on data in the European Registry of Quality Outcomes for Cataract and Refractive Surgery database. J Cataract Refract Surg. 2012;38:1086-1093
- Sachdev GS, Sachdev Mahipal. Optimizing outcomes with multifocal intraocular lenses. Indian Journal of Ophthalmology. 2017; 65(12):1294-1300
- Lichtinger A, Rootman DS. Intraocular lenses for presbyopia correction: past, present, and future. Curr Opin Ophthalmol. 2012; 23(1): 40-6
- Agarwal A, Narang P. Tooling up for glued IOLs: Significance of three-piece IOLs and injectors. Ocular Surgery News. 2016
- Acrysof IQ Monofocal IOL. Beye.com. https://www.beye.com/product/acrysof-iq-monofocal-iol. Accessed 5 Dec 2020
- Sensar Monofocal 3-piece Hydrophobic Acrylic IOL. IOLs.eu https://iols.eu/product/lenses/iols-for-aphakia/sensar-monofocal-3-piece-hydrophobic-acrylic-iol-ar40m-40me/ Accessed 5 Dec 2020
- Anterior Chamber IOL. Auralab. https://www.aurolab.com/aurolens-anterior-chamber-iol.asp Accessed 5 Dec 2020
- Calladine D, Evans JR, Shah S. Leyland M. Multifocal versus monofocal intraocular lenses after cataract extraction. Cochrane Database Syst. Rev. 2012; 9:CD003169
- Alfonso JF, Fernandez-Vega L, Puchades C, Montes-Mico R. Intermediate visual function with different multifocal intraocular lens models. J Cataract Refract Surg 2010; 36(5): 733-739
- Roach L, Shaw J. Eyes on Europe: New Options in Multifocal IOLs. EyeNet Magazine. December 2020.
- McAlinden C, Pesudovs K, Moore J. Measure of Quality of Vision: The Quality of Vision Questionnaire. Investigative Ophthalmology & Visual Science 2010; 51(5537-5545)
- Vilar C, Hida WT, deMedeiros A, Magalhaes K, Tzelikis P, Chaves M, Motta A, Carricondo P, Alves M, Nose W. Comparison between bilateral implantation of a trifocal intraocular lens and blended implantation of two bifocal intraocular lenses. Clin Ophthalmol 2017; 11: 1393-1397
- Monaco G, Gari M, Di Censo F, Poscia A, Ruggi G, Scialdone A. Visual performance after bilateral implantation of 2 new presbyopia-correcting intraocular lenses: trifocal versus extended range of vision. J Cataract Refract Surg 2017; 43(6): 737-747
- Kohnen T, Herzog M, Hemkeppler E, Schonbrunn S, DeLorenzo N, Petermann K, Bohm M. Visual Performance of a Quadrifocal (Trifocal) Intraocular Lens Following Removal of the Crystalline Lens. Amer J Ophthalmology; 2017; 184: 52-62.
- Kohnen T, Titke C, Bohm M. Trifocal intraocular lens implantation to treat visual demands in various distances following lens removal. Am J Ophthalmol 2016; 161: 71-77.
- Schwiegerling J, Petznick A.(2019) Refractive and Diffractive Principles in Presbyopia-Correcting IOLs—An Optical Lesson. [White Paper]. Alcon Medical Affairs. https://us.alconscience.com/sites/g/files/rbvwei1736/files/pdf/Refractive-and-Diffractive-Principles-in-Presbyopia-Correcting-IOLs-An-Optical-Lesson-US-REF-1900001.pdf
- Savini G, Schiano-Lomoriello D, Baducci N, Barboni P. Visual Performance of a New Extended Depth of Focus Intraocular Lens Compared to a Distance-Dominant Diffractive Multifocal Intraocular Lens. J Refract Surg. 2018; 34(4): 228-235
- FDA Summary of Safety and Effectiveness Data: Vivity Extended Vision IOL. FDA. https://www.accessdata.fda.gov/cdrh_docs/pdf/P930014S126B.pdf. 2020: 1-52
- Rubenstein JB, Raciti M. Approaches to corneal astigmatism in cataract surgery. Curr Opin Ophthalmol. 2013; 24:30-34
- Stodola E. Current and future IOL choices: Using toric IOLs to correct astigmatism. EyeWorld. March 2018. https://www.eyeworld.org/using-toric-iols-correct-astigmatism
- TECNIS Toric IOL Patient Information Brochure. https://www.accessdata.fda.gov/cdrh_docs/pdf/P980040S039c.pdf
- enVista Toric IOL (Model MX60T). https://www.bausch.com/ecp/our-products/cataract-surgery/lens-systems/envista-toric-iol
- Holland E, Lane S, Horn J, Ernest P, Arleo R, Miller K. The AcrySof Toric intraocular lens in subjects with cataracts and corneal astigmatism: a randomized, subject-masked parallel-group, 1-year study. Ophthalmology. 2010; 117(11):2104-11
- Lockhart TE, Shi W. Effects of Age on Dynamic Accomodation. Ergonomics. 2010; 53(7): 892-903
- Kent D. Accomodating IOLs: Two More Possibilities. Review of Ophthalmology. December 2019. https://www.reviewofophthalmology.com/article/accommodating-iols-two-more-possibilities
- Alio JL, Simonov A, Plaza-Puche AB, et a. Visual outcomes and accommodative response of the Lumina accommodative intraocular lens. Am J Ophthalmol. 2016; 164:37-48
- Hantera MM, Hamed AM, Fekry Y, Shoheib EA. Initial experience with an accommodating intraocular lens: controlled prospective study. J Cataract Refract Surg. 2010; 26(7):1167-1172
- FDA approves first implanted lens that can be adjusted after cataract surgery to improve vision without eyeglasses in some patients. FDA News Release. 2017. https://www.fda.gov/news-events/press-announcements/fda-approves-first-implanted-lens-can-be-adjusted-after-cataract-surgery-improve-vision-without
- Achieving customized vision with the light adjustable lens. https://www.rxsight.com/us/customizing-your-vision/ accessed 5 Dec 2020
- Summary of Safety and Effectiveness Data (SSED) Posterior Chamber IOL (IOL), UltraViolet (UV) Light Source/Light Adjustable Lens (LAL)/Light Delivery Device (LDD). https://www.accessdata.fda.gov/cdrh_docs/pdf16/P160055B.pdf
- Charman WN & Tucker J. The depth-of-focus of the human eye for Snellen letter. Am J Optom Physiol Opt. 1975: 52:3-21
- Kim WS, Park IK, Chun YS. Quantitative analysis of functional changes caused by pinhole glasses. Invest Ophthalmol Vis Sci. 2014; 55: 6679-6685
- Read L. The IC-8 IOL: Big Advantages Through Small Apertures. The Ophthalmologist. Sep 2019. https://theophthalmologist.com/subspecialties/the-ic-8-iol-big-advantages-through-small-apertures
- Oheineachiain R. Pinhole IOL: A New IOL that uses Pinhole effect Provides Increased Depth of Field. ESCRS Eurotimes. 6 Jul 2018
- Hilman Liz. Bringing Small Aperture Optics to the IOL Plane. ASCRS EyeWorld. April 2019
- Trindade CC, Trindade BC, Trindade FC, Werner L, Osher R, Santhiago MR. New pinhole sulcus implant for correction of irregular astigmatism. J Cataract Refract Surg. 2017; 43: 1297-1308
- IC-8 IOL. European Post-Market Study of AcuFocus IC-8 Small Aperture IOL Published. Eyewire News. 2017. https://eyewire.news/articles/european-post-market-study-of-acufocus-ic-8-small-aperture-iol-published/
- Agarwal P, Navon SE. Xtra focus pinhole IOL (Morchers GMBH) a novel approach to tackle irregular astigmatism and large pupillary defects with a single step surgery. BMJ Case Reports. 2019; 12(4):e228902