Diabetes leads to a plethora of health conditions including kidney disease, nerve damage, heart disease and vision loss.1 According to the International Diabetes Federation (IDF), diabetes remains one of the leading causes of preventable blindness in the working adult population.2
The disease affects 1 in 10 individuals in the US and more than 240 million individuals worldwide, where around one-third of patients will progress to develop retinopathy.3,4
Individuals with type 1 or type 2 diabetes can develop retinal complications including diabetic retinopathy (DR) and macular edema.3 The risk of developing diabetic retinopathy increases with the duration of diabetes.3
The retina is the only part of the entire human body where these changes can be non-invasively observed, which emphasizes the important role optometrists play in co-managing diabetes with primary care physicians and endocrinologists.4
Projections for diabetic retinopathy prevalence
A 2020 study suggested that approximately 103 million individuals were diagnosed with diabetic retinopathy during that year and about 28 million of these individuals had vision loss from advanced stages of retinopathy.2
Current studies project that by the year 2045, more than 160 million individuals will be diagnosed with diabetic retinopathy and vision-threatening stages of retinopathy is expected to affect over 44 million individuals worldwide.2
The importance of early detection in DR
These concerning trends have emphasized the importance of early identification and appropriate treatment and management of diabetic retinopathy to prevent blindness. Timely and early diagnosis, consistent disease classification, and stage-appropriate intervention can aid in the prevention of retinopathy and the prevention of advancing stages.5
According to the Centers for Disease Control and Prevention (CDC), about 90% of vision loss from diabetes can be prevented.6 Annual dilated eye examinations are essential for detecting retinopathy in its early stages and preventing progression.3,6
In addition, the integration of color fundus photography (CFP) and optical coherence tomography (OCT) into clinical practice supports more accurate staging of diabetic retinopathy and diabetic macular edema.3
The implementation of a grading system provides a clear framework for guiding both lifestyle modifications and medical treatment strategies, which leads to improved long-term visual outcomes and prevention of blindness.5
Diabetes can be associated with:3,6
- Cataracts
- Neovascular glaucoma
- Diabetic retinopathy
- Macular edema
- Dry eye disease
- Diabetic cranial nerve palsies
Grading diabetic retinopathy
DR occurs when the retinal arterioles, capillaries and venules become damaged from increased:3
- Oxidative stress
- Production of vascular endothelial growth factor (VEGF)
- Activation of the renin angiotensin system (RAS)
- Activity in metabolic pathways
Ultimately, chronically elevated blood glucose is associated with increased vascular permeability, retinal hemorrhages, lipid exudate and the formation of new blood vessels via increased VEGF expression.3
Diabetic retinopathy can be classified as non-proliferative or proliferative. Non-proliferative diabetic retinopathy (NPDR) presents with microaneurysms, intraretinal hemorrhages, venous beading, hard yellow exudates, cotton wool spots (soft exudates), and intraretinal microvascular abnormalities (IRMAs) as well as the absence of neovascularization.3,5,6 It can be further classified as mild, moderate, severe and very-severe based on clinical findings.3,5,6
Conversely, proliferative diabetic retinopathy (PDR) is associated with the addition of neovascularization of the optic disc (NVD) or elsewhere in the retina (NVE).5,6
Stages of DR
In addition, diabetic retinopathy is defined as either mild, moderate, severe, or very severe and is staged accordingly.
Stage 1: Mild NPDR
Mild NPDR is characterized by few microaneurysms and no other findings. Microaneurysms are outpouchings of retinal capillaries due to loss of vascular pericytes.3,6
Figure 1: Fundus image of a 70-year-old male with mild NPDR without macular edema in the right eye.
Figure 1: Courtesy of Jill Gottehrer, OD, FAAO.
Stage 2: Moderate NPDR
Moderate NPDR presents with at least one retinal hemorrhage or microaneurysms with the additional finding of at least one of the following:
- Intraretinal hemorrhages (dot and blot): Arise from leakage of the blood vessels that have weakened pericytes3,6
- Exudates: Yellow-white lipid-rich areas caused by chronic vascular damage and leakage5,7
- Cotton wool spots: Classified as “soft exudates” and appear as fluffy pale yellow or white areas; these are infarctions of the retinal nerve fiber layer of the retina5,7
- Venous looping or beading: Caused by severe retinal hypoxia and indicate an increased risk of progression to neovascularization5
Figure 2: Fundus photograph of moderate NPDR demonstrating microaneurysms and hemorrhages.
Figure 2: NPDR©International Centre for Eye Health. Image used under CC BY-NC 4.0.
Stage 3: Severe NPDR
Severe NPDR is characterized by either microaneurysms in all four quadrants, venous beading in at least two quadrants, or IRMA in one quadrant; this is known as the 4:2:1 rule.3,5-7
IRMAs are pre-existing vessels with proliferative endothelial cells or new blood vessel growth within the retina.3,5 These can be considered a precursor to neovascularization, therefore, in its presence, there is an increased risk of PDR.3,5
Figure 3: Fundus photographs of severe NPDR OU in a 63-year-old female with obstructive sleep apnea (OSA).
Figure 3: Courtesy of Kevin Cornwell, OD.
Stage 4: Very vevere NPDR
In short, very severe NPDR consists of two or more categories of the 4:2:1 rule.3,5
Figure 4: Fundus photograph of very severe NPDR demonstrating cotton wool spots, hemorrhages, and microaneurysms.
Figure 4: NPDR©International Centre for Eye Health. Image used under CC BY-NC 4.0.
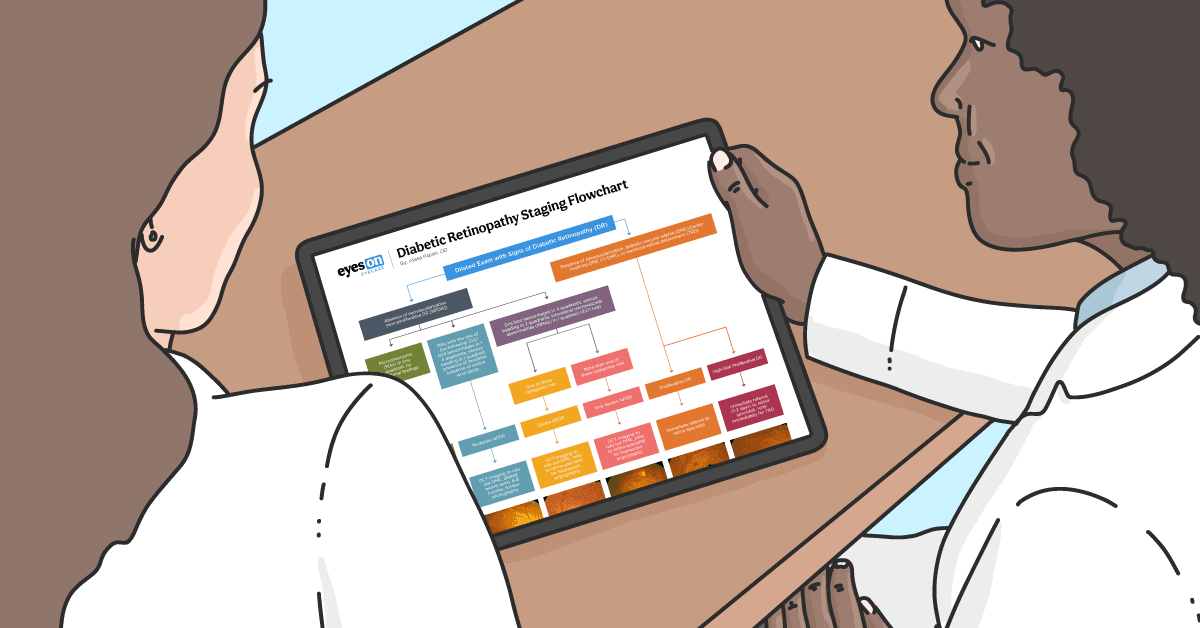
Download the flowchart here!

Diabetic Retinopathy Staging Flowchart
Use this flowchart to aid with monitoring DR patients and providing appropriately-timed referrals to enhance patient care.

Stage 5: Proliferative DR
Proliferative DR is a response of chronic ischemia and presents with clinical signs of NPDR at any stage with the addition of neovascularization (NV) at the optic disc or other areas of the retina.3,5,6
These new blood vessels are leaky and can be associated with a preretinal and subhyaloid vitreous hemorrhage.3,5 Additional complications of proliferative DR include tractional retinal detachment (TRD) and fibrovascular proliferation.3,5
Figure 5: Active neovascularization in PDR. Fibrovascular proliferation overlies the optic disc (white arrow). Loops of new vessels are especially prominent superior to the disc and extending into the macula, where leakage of fluid has led to deposition of a ring of hard exudate around the neovascular net (black arrow).
Figure 5: PDR©UCSF Department of Ophthalmology. Image used under CC BY-NC-ND 2.0.
Stage 6: High-risk PDR
High-risk PDR is characterized by the presence at least three of the four risk factors for severe visual loss from diabetic retinopathy, including the presence of:8
- Pre-retinal or vitreous hemorrhage
- New vessels
- New vessels on or near the disc (NVD)
- Moderate or severe new vessels (NV ≥standard photograph 10A or NVE ≥1/2 disc area [DA])
Figure 6: Fundus photography demonstrating scattered dot/blot hemorrhages OU, NVD OD>>OS, and NVE OU with fibrotic changes OD with epiretinal membrane.
Figure 6: Courtesy of Robert Africano, OD, FAAO.
Classifying diabetic macular edema
Diabetic macular edema (DME) is characterized by retinal thickening involving the macula (with or without lipid exudates or cystoid changes) due to diabetes,8 and is the leading cause of moderate to severe vision loss in diabetics.3,8 Of note, DME may occur at any stage of diabetic retinopathy and therefore, should be evaluated independently.3,8
This condition can be further classified into center-involving (CI-DME) and non-center-involving DME (NCI-DME) using OCT imaging of the central subfield thickness (CST), which is the central 1mm diameter of the fovea.8
DME is classified as follows:
- Non-center-involving DME (NCI-DME): Affects the area outside the CST zone, therefore most patients retain the central or “straight ahead” vision with this form8
- Center involving DME (CI-DME): Considered more visually debilitating as this form involves the CST and directly impacts the patient’s central vision8
- Clinically significant macular edema (CSME): DME is considered CSME if there is:
- Retinal thickening of 500µm (⅓ disc diameter [DD]) or less from the macula3,5,6,8
- Hard exudates 500µm (⅓ DD) or less from the center of the macula with adjacent retinal thickening or one or more zones of retinal thickening of at least 1 DD in size and within 1 DD from the center of the fovea3,5,6,8
- Note, the use of the terms CI-DME and NCI-DME have largely replaced CSME in the grading of DME8
Accurate classification of DME, particularly distinguishing center-involving from non-center-involving disease, is essential for guiding management and optimizing visual outcomes.3,8
Figure 7: Fundus photograph of moderate NPDR with CI-DME.
Figure 7: Courtesy of Inrava Khasnabish, OD, FAAO.
Figures 8 and 9: Fundus photography OD and OS, respectively, of a 51-year-old male with PDR and DME.
Figure 8: Courtesy of Jill Gottehrer, OD, FAAO.
Figure 9: Courtesy of Jill Gottehrer, OD, FAAO.
Treating and managing diabetic retinopathy and macular edema
According to the American Optometric Association (AOA), a patient recently diagnosed with type 1 diabetes should have an eye examination within the first 5 years of diabetic onset, whereas an individual with type 2 diabetes should be evaluated at the time of diagnosis.5,9
For type 1 and 2 diabetes without retinopathy, dilated examinations are recommended and these patients can bee seen every 1 to 2 years.9 Patient education on the importance of glycemic control, including medical adherence and essential lifestyle modifications is important to address in the presence or absence of retinopathy.2,3
Mild NPDR
Individuals diagnosed with mild NPDR without DME can be monitored annually with dilated eye examinations.3,5 If a patient has CI-DME, they should be referred within 2 to 4 weeks, whereas NCI-DME can be managed more conservatively if the patient maintains good visual acuity.3,6,9 Referral would be warranted if the edema is progressing, if vision is worsening, or if the clinician is unsure.3,6,9
OCT is the primary modality used to diagnose and monitor DME.3 Fluorescein angiography may be used selectively to evaluate areas of leakage or macular ischemia but is not routinely required.3 Management is guided by OCT findings, particularly the presence of center-involving DME.
First-line treatment is typically intravitreal anti-VEGF therapy, while focal/grid laser photocoagulation has a limited, adjunctive role, primarily in NCI-DME, and is not used preventatively in eyes without edema.3,6
Moderate NPDR
Moderate NPDR should be monitored every 6 to 8 months as up to approximately 12 to 27% of cases advance to PDR within 1 year.5 The use of fundus imaging is helpful in monitoring for progression of disease.3,5 In all cases of NPDR, OCT is recommended to rule out DME.3
Severe and very severe NPDR
Severe NPDR and very severe NPDR should be co-managed with a retinal specialist and followed every 3 to 4 months and even more frequently if very severe.3,5 Referral to ophthalmology for closer monitoring with OCT and fluorescein angiography is recommended as the risk of vision loss and PDR increases significantly at this stage.5
It is important to update the patient’s PCP or endocrinologist of clinical findings as organ damage at other parts of the body may be present.5
Proliferative DR
Proliferative DR should be referred to retina within 1 week if not high risk, within 1 to 2 days if high risk, and immediately if there is evidence of a tractional RD.3,5,6,9 Panretinal laser photocoagulation (PRP) is the classified as the standard care for peripheral neovascularization.3,5
According to a study, PRP reduces the risk of severe vision loss in PDR from 16% to less than 7%.3 It is important to note that PRP and other forms of laser treatment do not improve visual acuity; it instead prevents the advancement of retinopathy and progression of vision loss.3
Anti-VEGF injections may also be indicated in conjunction with PRP in the treatment of PDR and have shown to also improve visual acuity.3,5,8
Red flags to note in diabetic retinopathy management:3,8,9
- Moderate to severe NPDR (does not require immediate attention)
- Suspicion of diabetic macular edema and/or clinically significant macular edema (does not require immediate attention)
- Presence of neovascularization
- Presence of pretinal and subhyaloid hemorrhage
- Presence of tractional retinal detachment
In a rush? Click here to download the Diabetic Retinopathy Staging Flowchart!
Deeper dive into interventions for PDR and DME
Anti-VEGF agents
There are five intravitreal anti-VEGF agents used in the management of PDR, including:3,5
- Aflibercept 2mg (EYLEA),
- Aflibercept 8mg (EYLEA HD)
- Bevacizumab (AVASTIN)
- Ranibizumab (LUCENTIS)
- Faricimab (VABYSMO)
Aflibercept has a higher affinity to VEGF and has a longer duration of action compared to other agents.3 Bevacizumab is used off label in the treatment of diabetic retinopathy and is associated with higher recurrence and lower effectivity compared to aflibercept.3
Faricimab inhibits both VEGF-A and angiopoietin-2 (Ang-2), which may confer additional benefits by stabilizing the vasculature and reducing leakage. Anti-VEGF injections can treat PDR and dissect fibrovascular membranes associated with PDR.3
There is a small but recognized risk of TRD shortly after anti-VEGF injection in eyes with PDR, particularly in those with significant fibrovascular proliferation; therefore, close follow-up in the early post-treatment period is recommended.5
Monitoring and co-managing DME
In eyes with DME, treatment is guided by OCT findings and whether the edema is center-involving. CI-DME with vision impairment is typically treated with intravitreal anti-VEGF therapy as first-line.5 Intravitreal corticosteroids may be considered in selected cases (e.g., inadequate response or contraindications to anti-VEGF).
Focal or grid laser photocoagulation has a limited, adjunctive role and is primarily used for NCI-DME or persistent focal leakage. Follow-up is generally performed at regular intervals (often every 4 to 12 weeks initially) based on clinical response and OCT findings.5
Fluorescein angiography may be used selectively to evaluate leakage or macular ischemia but is not routinely required for retreatment decisions.
Intravitreal steroids
Intravitreal steroids, such as injections of triamcinolone acetonide (IVTA) and slow-releasing dexamethasone implants (OZURDEX, AbbVie), possess both anti-inflammatory and anti-angiogenic properties, which inhibits VEGF production.5
Consequently, these agents are effective in the treatment of DME, particularly in pseudophakes or in those that do not get an adequate treatment response with anti-VEGF agents. Intravitreal steroid injections are maximally effective for approximately 2 to 4 months, while OZURDEX has a longer duration of action (3 to 6 months).3,8
Risks associated with intravitreal steroids include elevated IOP and development of cataract.3,8 OZURDEX is a slow-releasing dexamethasone implant which may reduce the risk of cataract and glaucoma in some patients.3
Pars plana vitrectomy
Pars plana vitrectomy (PPV) or enzymatic vitrectomy is indicated in individuals with PDR complicated by non-clearing vitreous hemorrhage, tractional retinal detachment or combined tractional-rhegmatogenous retinal detachment.3,8
It is also considered in select cases of DME when there is a tractional component or when the DME is not responding to conventional therapy.3,8
Key takeaways
- Diabetic retinopathy is one of the leading causes of blindness worldwide2
- Approximately 25% of patients with moderate NPDR will progress to PDR within 1 year5
- DME and PDR should be referred for prompt treatment with a retina specialist3,5,6,9
- Fundus photography, OCT imaging, and fluorescein angiography aid in appropriate grading of the disease3,5
- Clinically significant macular edema (CSME) represents vision-threatening diabetic macular edema and can lead to central vision loss if untreated8
Conclusion
The risk of developing diabetic retinopathy increases with the duration of diabetes, making regular ocular monitoring essential in preventing blindness from the disease.3 Effective management requires not only accurate staging of retinopathy and identification of DME but also coordinated care with the patient’s primary care physician and/or endocrinologist.5
Co-management ensures systemic factors such as glycemic control and hypertension are addressed alongside ocular interventions, which is critical in preventing vision loss and other diabetes-related complications.3,5 Patient education remains a cornerstone of care; individuals should understand their clinical findings, the importance of adherence to treatment, and lifestyle modifications, including glycemic and blood pressure control.5
Standardized grading and consistent use of diagnostic tools, including fundus photography, OCT, and fluorescein angiography, allow for precise monitoring, timely referrals, and stage-appropriate interventions.3,5
A multidisciplinary approach that integrates patient education, systemic disease management, and advanced ocular care optimizes visual outcomes and reduces the risk of preventable blindness in diabetic patients.5