Age-related macular degeneration (AMD) is one of the retinal conditions that we all learn about early on in our optometric education. AMD seems so ubiquitous that it is often underappreciated or even worse, unseen. In fact, though over 3.6 million people are projected to have AMD by 2030 in the USA alone, a recent study has shown that 25% of AMD in the primary eyecare setting goes undiagnosed.1,2 As with all things, our understanding of AMD is quickly advancing. Although I have tried my hardest to include all the pertinent AMD information into one article, I’ve never been known for my brevity. So consider this a quick refresher before we move on to more detailed analyses of AMD patients.
Age-related macular degeneration is a macular deterioration, most often occurring in patients over 50 years of age, characterized by drusen, pigmentary abnormalities, geographic atrophy, and/or choroidal/macular neovascularization, not explained by another retinal condition. Since neovascularization within the macula can originate from the choroidal or retinal circulation, the term choroidal neovascularization has been replaced with a less specific term, macular neovascularization (MNV). There are three types of MNV (summarized in Table 1), all of which can lead to subretinal fluid, intraretinal fluid, hemorrhage, lipid exudation, and/or fibrosis.
Table 1
| AMD Finding | Description |
|---|
| Drusen | Yellow-white focal deposits of extracellular debris |
| Pigment Abnormalities | Focal area of increased or decreased pigmentation, including small areas of RPE loss |
| Geographic Atrophy | Well demarcated RPE atrophy leading to improved visualization of the underlying choroid, may be associated with choriocapillaris and/or outer retinal atrophy |
| Exudative MNV | Pathological macular angiogenesis associated with hemorrhage, subretinal fluid, intraretinal fluid, and/or lipid exudation |
| Nonexudative MNV | Pathological macular angiogenesis without associated hemorrhage, subretinal fluid, intraretinal fluid, or lipid exudation |
| Type 1 MNV | Pathological macular angiogenesis, originating from the choriocapillaris, within the subRPE space |
| Type 2 MNV | Pathological macular angiogenesis, originating from the choriocapillaris, within the subretinal space |
| Type 3 MNV | Pathological macular angiogenesis, originating from the retinal circulation, often grows towards the RPE |
- RPE: retinal pigment epithelium
- MNV: macular neovascularization
“Drusen” is a catchall term for yellow-white round lesions noted on funduscopy that are thought to represent extracellular accumulations of debris. Drusen are not unique to AMD or even to the macula: they can be isolated findings or associated with other retinal conditions throughout any portion of the retina. AMD patients with findings of drusen only most often have good vision, though central drusen with overlying atrophy may reduce visual acuity or cause metamorphopsia. The primary concern with drusen is that they confer a risk of conversion to late AMD, which often significantly impacts visual function.
Table 2
| Finding | Description |
|---|
| Soft Drusen 3 | Extracellular deposits between Bruch’s membrane and RPE with mound-like appearance, can be confluent, range in size from >63μm to >1000μm in base diameter |
| Cuticular Drusen3 | Extracellular deposits between Bruch’s membrane and RPE with blunted triangle appearance, often have apical RPE thinning, range in size from >50μm to 75μm in base diameter |
| Subretinal Drusenoid Deposit3 | Extracellular deposits anterior to the RPE with varying shape, ranging from 25μm to >1000μm in base diameter |
| Small Drusen/Drupelets4 | Drusen <63μm in base diameter |
| Medium/Intermediate Drusen 4 | Drusen ≥63μm and ≤125μm in base diameter |
| Large Drusen4 | Drusen >125μm in base diameter |
Subretinal drusenoid deposits or reticular pseudodrusen (as they were previously called) are clinically and histologically similar structures to drusen. Unlike drusen, which form between Bruch’s membrane and the RPE, subretinal drusenoid deposits are found anterior to the RPE. Both drusen and subretinal drusenoid deposits confer risk of vision loss and progression to advanced stages of AMD. Drusen and subretinal drusenoid deposits are often transient structures that can change size, shape, and even resorb. Drusen resorption is often the harbinger of outer retinal atrophy, vision loss, or advanced AMD, therefore these patients should be monitored more closely.
In Image 1, we see the three types of drusen. Note the relative position of the RPE and druse structure.
Image 1: Three types of drusen. Note the relative position of the RPE and druse structure.
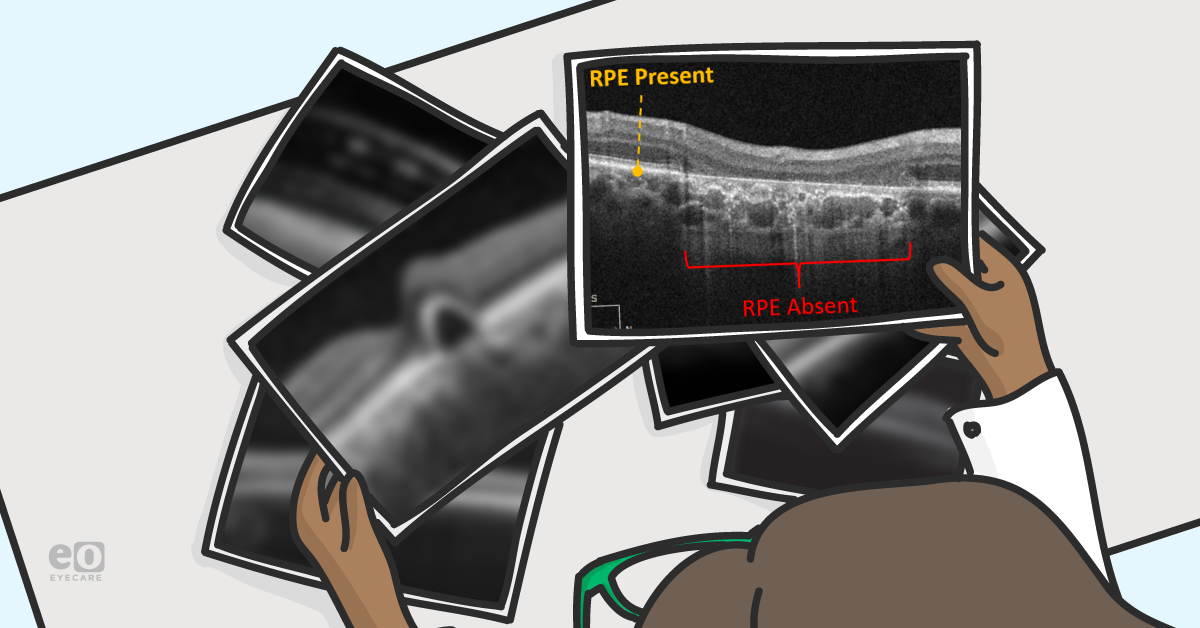
Geographic atrophy is the loss of the RPE monolayer. These lesions appear as well-demarcated ovoid areas of hypopigmentation with increased visualization of the underlying choroid. Patients will often have co-localizing outer retinal and/or choriocapillaris atrophy as well. When visualized with OCT, the RPE will be absent with collapse of the overlying retina. The choroid will be relatively hyper-reflective and seen in greater detail due to the loss of the RPE’s masking effect, which absorbs much of the OCT signal. Patients will have a slowly progressive loss of vision and though there are no currently well-accepted treatments for geographic atrophy, these patients must still be monitored for conversion to neovascular AMD, which may require treatment.
Image 2 shows geographic atrophy with overlying outer retinal atrophy. Note the increased reflectance and visualization of the underlying choroid.
Image 2: Geographic atrophy with overlying outer retinal atrophy. Note the increased reflectance and visualization of the underlying choroid.
Neovascular AMD is a late form of AMD characterized by pathological macular angiogenesis, hemorrhage, subretinal fluid, intraretinal fluid, and/or exudation. The classic form of neovascular AMD is exudative MNV which presents with hemorrhage, fluid, and/or lipid exudation. When present within the fovea, exudative MNV can lead to profound vision loss over the course of weeks to months. Fortunately, the advent of anti-VEGF therapy has significantly improved the visual outcome in these patients, with stabilization and even remediation of vision loss with prompt treatment.
With the
emergence of OCT-A, a new form of neovascular AMD has been defined: nonexudative MNV. Nonexudative MNV is a neovascular lesion without fluid and causes minimal disruption to retinal architecture and vision. While exudative MNV has numerous randomized controlled trials to dictate treatment, the management of exudative MNV is far less standardized and is more case-specific.
Look out for the next issue which will delve deeper into both forms of neovascular AMD!
In Image 3, exudative macular neovascularization with subretinal fluid, intraretinal fluid, and lipid exudation is represented.
Image 3: Exudative macular neovascularization with subretinal fluid, intraretinal fluid, and lipid exudation.
When diagnosing AMD, it is important to grade the disease severity accurately to help facilitate proper management and discussion with other health care providers. Table 3 summarizes two commonly used AMD staging systems: the AREDS classification and the Beckman Committee classification. These classifications are clinical (or fundus color photographic) scales that categorize phenotypic presentations of each individual eye, to allow for standardized risk stratification and treatment.
Table 3
| AMD Stage | Beckman Committee Criteria* | AREDS Criteria |
|---|
| No apparent aging changes | No drusen and no pigment abnormalities | ― |
| Normal aging changes | Small drusen/drupelets only | ― |
| No AMD | ― | No or few small drusen |
| Early AMD | Medium drusen only | Multiple small drusen, few medium drusen, and/or mild RPE abnormalities |
| Intermediate AMD | Large drusen and/or pigment abnormalities | Numerous medium drusen, at least 1 large druse, and/or non-center involving geographic atrophy |
| Late/Advanced AMD | Neovascular AMD and/or geographic atrophy | Center involving geographic atrophy, MNV or current/past signs of MNV |
*All findings must be within 2 disc diameters of the fovea
Drusen: includes any drusen type and subretinal drusenoid deposits
Pigment abnormalities: any hyperpigmentation or hypopigmentation associated with medium or large drusen
RPE: retinal pigment epithelium
MNV: macular neovascularization
As summarized in Table 4, below, management is based on the stage of AMD, type of AMD, and patient specific risk factors. This management is my personal and general approach to AMD, and pulls from the American Optometric Association and American Academy of Ophthalmology preferred practice patterns. As with all management guidelines, it is important to use them as a model and not strict criteria.
My approach to AMD and to all retinal disease is to monitor closely after initial diagnosis and then slowly increase the follow up period as the individual’s disease state and risk factors dictate. I always think about “what if”: I can’t cause any significant patient harm if I see the patient back for an extra follow up, but I may cause irreversible vision loss if I miss something by setting a long follow up interval.
All patients should be counseled on risk modification, both systemic and environmental. Patients with signs of AMD should all monitor vision with either a monocular visual self-assessment, Amsler grid, or a Foresee Home system. Though monocular visual self-assessment and Amsler grids are not highly sensitive in identifying AMD-related visual function decreases, they are low-tech and cheap ways for patients to evaluate their own vision. The Foresee Home is an electronic system based on Vernier acuity that allows patients to assess their vision with high sensitivity and has been shown to improve visual outcomes in patients who develop MNV.
Table 4
| AMD Type | Follow-up* | Additional Management |
|---|
| Normal aging changes | 12 months | Counsel on proper diet and exercise, smoking cessation, ocular UV protection |
| Early AMD | 6-12 months | Counsel on proper diet and exercise, smoking cessation, ocular UV protection, and vision self-assessment |
| Intermediate AMD | 4-6 months# | • AREDS2 formulation • Counsel on proper diet and exercise, smoking cessation, ocular UV protection, and vision self-assessment |
| Exudative Neovascular AMD | Refer for Treatment | • AREDS2 formulation if fellow eye does not have neovascular AMD or significant central geographic atrophy • Counsel on proper diet and exercise, smoking cessation, ocular UV protection, and vision self-assessment • Consider low vision evaluation |
| Nonexudative Neovascular AMD | Refer for treatment or monitor 1-3 months^ | • AREDS2 formulation if fellow eye does not have neovascular AMD or significant central geographic atrophy • Counsel on proper diet and exercise, smoking cessation, ocular UV protection, and vision self-assessment |
| Geographic Atrophy | 6-12 months# | • AREDS2 formulation if fellow eye does not have neovascular AMD or significant central geographic atrophy • Counsel on proper diet and exercise, smoking cessation, ocular UV protection, and vision self-assessment • Consider low vision evaluation |
*Author’s best practices, see American Optometric Association and American Academy of Ophthalmology for practice guidelines
#Consider monitoring more closely if fellow eye has/had CNV or if patient is monocular
^Requires cautious observation with a multimodal imaging approach (at least OCTA and structural OCT)
In conclusion
We will all see AMD in our daily clinical lives. Unfortunately,
the number of patients with AMD will continue to grow as our population ages, but newer technology and research is allowing us to personalize our approach to AMD. We are at the precipice of personalized medicine, where we can combine the latest technology with research expertise to identify novel risk factors to guide our management of AMD patients. Ultimately, this will help us recognize those patients who need to be followed more closely while alleviating the fears of our low-risk AMD patients. Join me in our next issue as we delve a little bit deeper into macular neovascularization.
Further reading
- https://www.nei.nih.gov/learn-about-eye-health/outreach-campaigns-and-resources/eye-health-data-and-statistics/age-related-macular-degeneration-amd-data-and-statistics/age-related-macular-degeneration-amd-tables
- Neely DC, Bray KJ, Huisingh CE, et al. Prevalence of undiagnosed age-related macular degeneration in primary eye care. JAMA Ophthalmol. 2017;135(6):570-5.
- Spaide, Richard F., and Christine A. Curcio. "Drusen characterization with multimodal imaging." Retina (Philadelphia, Pa.) 30.9 (2010): 1441.
- Ferris III, Frederick L., et al. "Clinical classification of age-related macular degeneration." Ophthalmology 120.4 (2013): 844-851.