There is the mild itchy eye due to allergies, and then there is vernal keratoconjunctivitis (VKC). VKC is a pronounced ocular response to allergens, often with significant reactions involving the conjunctiva, limbus, and cornea. It typically affects school-aged males,1 however there can be a similar presentation in adults known as atopic keratoconjunctivitis (AKC). VKC can present year-round, with seasonal flare-ups, and has been associated with comorbid allergic conditions or family history of atopy.2
Zooming in on the cellular level
The clinical characteristics of VKC come from a complex allergic cascade. It has primarily been attributed to IgE, which triggers the release of histamine and other mast cell mediators. These players are built into the immune system; however, it is the exaggerated response to common allergens that manifests as VKC. Eosinophils also contribute to the pathogenesis of VKC and have been considered a marker of the disease.2-5 Besides allergic mechanisms, innate immunity, hormones, and neuroinflammation also seem to play a role in VKC.2
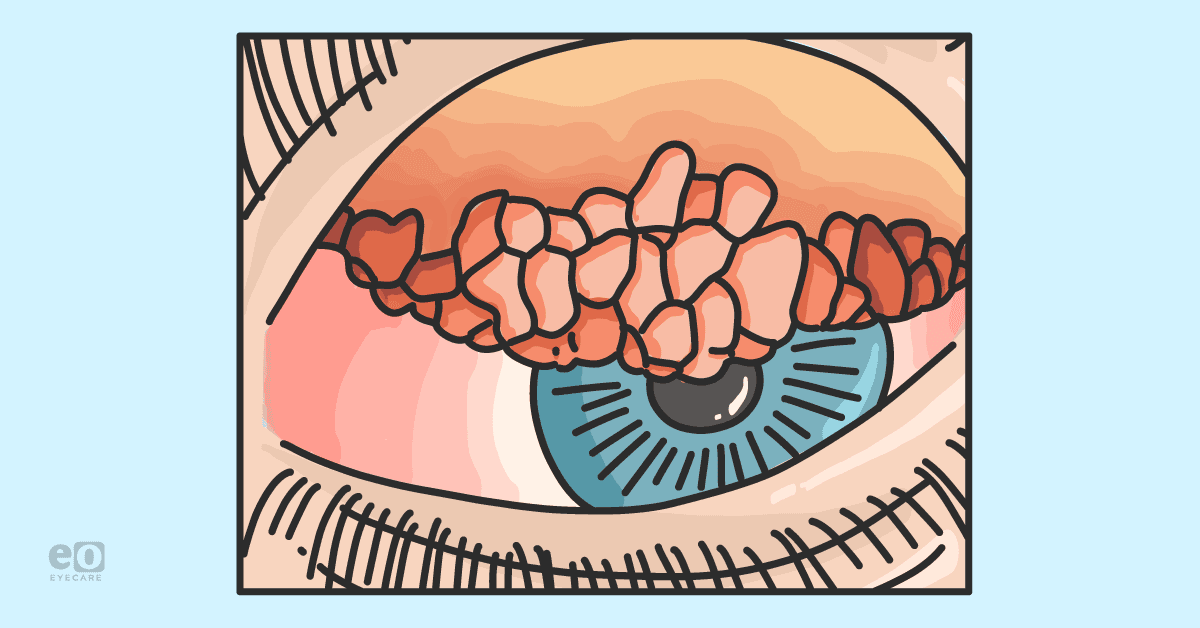
Clinical picture of VKC
To reiterate, VKC goes beyond simple allergic conjunctivitis, as it is a hypersensitivity reaction to allergens. Presentation can be variable, including some or all of the characteristic signs. Symptoms include significant foreign body sensation, redness, swelling, tearing, and sometimes blurred vision.
| Location | Findings |
|---|---|
| Conjunctiva | Giant papillae: The papillae seen in VKC are hard to miss. Even from outside of the slit lamp, they are prominent, cobblestone-like papillae on the superior tarsal conjunctiva. |
| Discharge: The discharge of VKC is typically thick and ropy. | |
| Cornea | Superficial punctate keratopathy: The epithelium can be rough locally or diffusely from mechanical injury from the papillae. When untreated or severe, these erosions are the precursor to a shield ulcer.6 |
| Shield ulcer: Toxic proteins excreted from conjunctival eosinophils break down the epithelium and form these oval plaques.7 They are usually found in the superior part of the cornea in severe VKC. | |
| Corneal scarring: Scars are a result of recalcitrant shield ulcers or limbal stem cells damage.7 | |
| Limbus | Horner-Trantas dots: Confluent, gelatinous papillae can have eosinophilic concretions and a white-capped appearance circumscribing the limbus. |
Therapeutic approaches: The tried and true
Because of its chronicity and underlying atopic mechanisms, VKC requires short-term and long-term management. To address acute symptoms, first-line standard of care is usually topical dual-acting mast cell stabilizers and antihistamines, such as olopatadine and ketotifen. These drops target the root cause (mast cell degranulation) and the effect (symptoms from histamine) in VKC, and have been shown to relieve most of its signs and symptoms.
For VKC, which follows predictable seasonal patterns, these dual-acting drops can be initiated in anticipation of their symptoms and throughout the season. Oral antihistamines can be considered as adjunct therapy in this first stage of treatment.
When there is more severe disease and the cornea is still intact, topical corticosteroids should be utilized to reduce inflammation. Before reaching for prednisolone acetate 1% which can have more significant side effects, consider fluorometholone 0.1%. A short course of flurometholone 0.1%, dosed one drop four times daily, can mitigate much of the ocular discomfort associated with VKC.8
Immunomodulators have been used in VKC unresponsive to first-line therapies and for long-term management. Cyclosporine is not new in the treatment of VKC, and cyclosporine at 0.05%9 and 2%10 concentrations is an option as a steroid-sparing agent for compromised corneas. Cyclosporine may be familiar in different strengths as the brand-name drugs Restasis (0.05%) and Cequa (0.09%). Tacrolimus, another immunomodulator, has been shown to be effective at 0.1%, 0.005%, and 0.03% concentrations for VKC as well.11 Both cyclosporine and tacrolimus work by blocking the release of pro-inflammatory cytokines.
Therapeutic approaches: The new
As of the summer of 2021, Santen’s Verkazia (cyclosporine ophthalmic emulsion 0.1%) was approved by the FDA for the treatment of VKC in children ages 4 to 18. Where does Verkazia fit into the current management of VKC? As for any new-to-market medications, there are many factors to thoughtfully consider before prescribing Verkazia for your patient with VKC.
Efficacy and safety
FDA-approved medications must undergo rigorous clinical trials before they are available to be prescribed. The NOVATIVE12 and VEKTIS13 studies demonstrated Verkazia’s success when dosed four times daily for severe VKC. It improved symptoms and corneal signs after as early as 1 month of use and had sustained effects for a 12-month period.14
Verkazia was more effective than the lower 0.05% concentration already commercially available, which would be used off-label.12 It is considered safe with minimal side effects or adverse events, which is obviously very important for the FDA but also for parents who will be administering this medication to their children.
Cost
A patient’s access to a brand-name drug can be largely affected by the cost of the drug and the drug’s inclusion on prescription drug formularies. Because Verkazia is so new, its US pricing and drug coverage is unavailable; however, when compared to the low cost of generic or other common branded allergy drops and steroid drops, Verkazia comes out to be up to 35 times more expensive.15 At the time this article was authored, Santen did not have a patient assistance program for this medication.
In conclusion
VKC is a chronic disease of hypersensitivity to allergens, with potentially visually debilitating consequences. The most severe forms of VKC can involve the conjunctiva, cornea, and limbus, while milder forms can be limited to the conjunctiva alone. Initial therapy usually includes a topical dual-acting mast cell stabilizer and antihistamine, and a short course of corticosteroids can be added for more serious cases. Topical immunomodulators are generally reserved for refractory VKC, now with Verkazia as a promising FDA-approved option.
References
- Leonardi A, Busca F, Motterle L et al. Case series of 406 vernal keratoconjunctivitis patients: a demographic and epidemiological study. Acta Ophthalmol Scan. 2006 Jun;84(3):406-10.
- Bonini S, Bonini S, Lambiase A et al. Vernal keratoconjunctivitis revisited: a case series of 195 patients with long-term followup. Ophthalmology. 2000 Jun;107(6):1157-63.
- Sachet M, Plateroti R, Bruscolini A et al. Understanding Vernal Keratoconjunctivitis: Beyond Allergic Mechanisms. Life (Basel). 2021 Sep 26;11(10):1012.
- Bruschi G, Ghiglioni DG, Osnaghi S et al. Role of ocular cytology in vernal keratoconjunctivitis. Immun Inflame Dis. 2020 Mar;8(1):3-7.
- Irkec MT, Bozkurt B. Molecular immunology of allergic conjunctivitis. Curr Opin Allergy Clin Immunol. 2012 Oct;12(5):534-9.
- Feizi S, Javadi MA, Alemzadeh-Ansari M et al. Management of corneal complications in vernal keratoconjunctivitis: a review. Ocul Surf. 2021 Jan;19:282-289.
- Solomon A. Corneal complications of vernal keratoconjunctivitis. Curr Opin Allergy Clin Immunol. 2015 Oct;15(5):489-94.
- Tabbara KF, al-Kharashi SA. Efficacy of Nedocromil 2% versus flurometholone 0.1%: a randomized, double masked trial comparing the effects on severe vernal keratoconjunctivitis. Br J Ophthalmol. 1999 Feb;83(2):180-4.
- Chatterjee A, Bandyopadhyay S, Bandyopadhyay SK. Efficacy, Safety and Steroid-sparing Effect of Topical Cyclosporine A 0.05% for Vernal Keratoconjunctivitis in Indian Children. J Ophthalmic Vis Res. 2019 Oct 24;14(4):412-418.
- Pucci N, Novembre E, Cianferoni A et al. Efficacy and safety of cyclosporine eyedrops in vernal keratoconjunctivitis. Ann Allergy Asthma Immunol. 2002;89(3):298.
- Zhao M, He F, Yang Y et al. Therapeutic efficacy of Tacrolimus in vernal keratoconjunctivitis: a meta-analysis of randomize controlled trials. Our J Host Pharm. 2020 Nov 3.
- Amrane M, Bremond-Gignac D, Baudouin C et al. A multicenter, double-masked, randomized, parallel group, controlled trial of efficacy and tolerance of NOVA22007 (cyclosporine cationic emulsion) versus vehicle in patients with vernal keratoconjunctivitis. IOVS. 2011 52:6415.
- Leonardi A, Doan S, Amrane M et al. A Randomized, Controlled Trial of Cyclosporine A Cationic Emulsion in Pediatric Vernal Keratoconjunctivitis: The VEKTIS Study. Ophthalmology. 2019 May;126(5):671-681.
- Bremond-Gignac D, Doan S, Amrane M et al. Twelve-Month Results of Cyclosporine A Cationic Emulsion in a Randomized Study in Patients with Pediatric Vernal Keratoconjunctivitis. Am J Ophthalmol. 2020 Apr;212:116-126.
- Pharmacoeconomic Review Report: Cyclosporine (Verkazia): (Santen Canada Inc.) Ottawa (ON): Canadian Agency for Drugs and Technologies in Health; 2020 Jan.